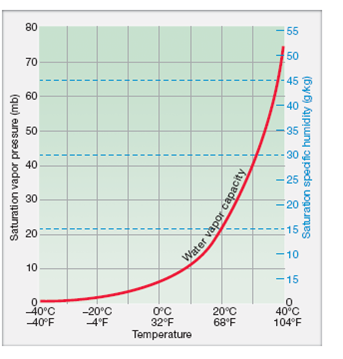
See also other properties of Ethylene at varying temperature: Dynamic and kinematic viscosity, Specific heat (heat capacity) and Thermal conductivity, and Thermophysical properties at standard conditions ,Īs well as density and specific weight o f acetone, air, ammonia, argon, benzene, butane, carbon dioxide, carbon monoxide, ethane, ethanol, ethylene, helium, hydrogen, methane, methanol, nitrogen, oxygen, pentane, propane, toluene and water. Note that there will be a significant shift in density when going from liquid to gaseous phase of ethylene (below and above boiling point at given pressure)Ĭhoose the actual unit of temperature: ☌ ☏ K °R Choose the actual pressure: 1 bara / 14.5 psia 10 bara / 145 psia 50 bara / 725 psia Specific weight is given as N/m 3 and lb f / ft 3. 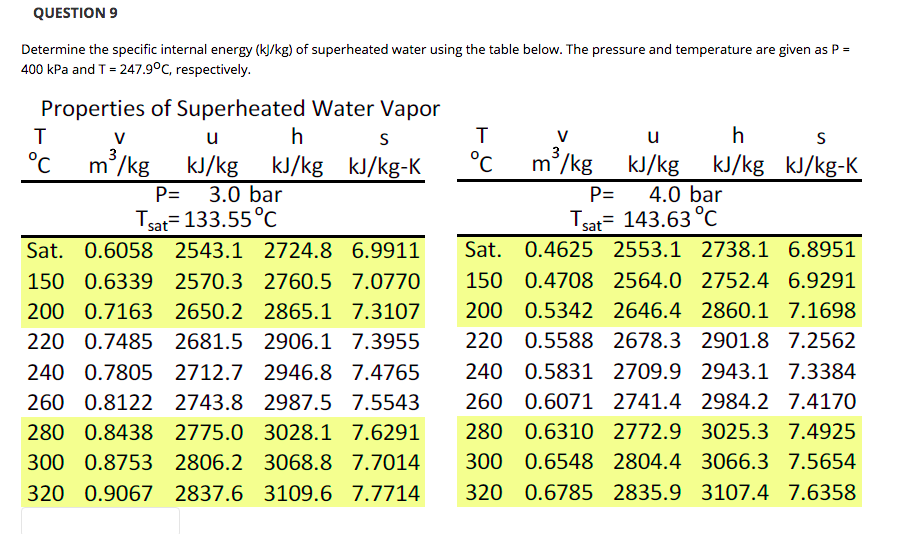
The output density is given as kg/m 3, lb/ft 3, lb/gal(US liq) and sl/ft 3. The calculator below can be used to estimate the density and specific weight of ethylene at given temperature and pressure. Tabulated values of ethylene density and specific weight at given temperature and pressure (SI and Imperial units) as well as density units conversion are given below the figures. Where g = acceleration due to gravity, units typically and value on Earth usually given as 9.80665 or 32.17405 Specific weight, γ, has units typically or is defined by the ratio of the weight to the volume of a substance: 
Density, ρ, has units typically or, and is defined by the ratio of the mass to the volume of a substance:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
